Innovatieve hardware voor de zorg
Easy Press
Gemakkelijke instap, lage artrogene belasting en maximale aanpassing van spieren zijn de belangrijkste kenmerken van de Monitored Rehab Systems Easy Press. De Easy Press is bij uitstek geschikt voor de oudere patiënt.
De Easy Press biedt de unieke mogelijkheid tot enkel- of dubbelzijdig trainen, begeleid door de revolutionaire MRS Software voor visuele feedback.
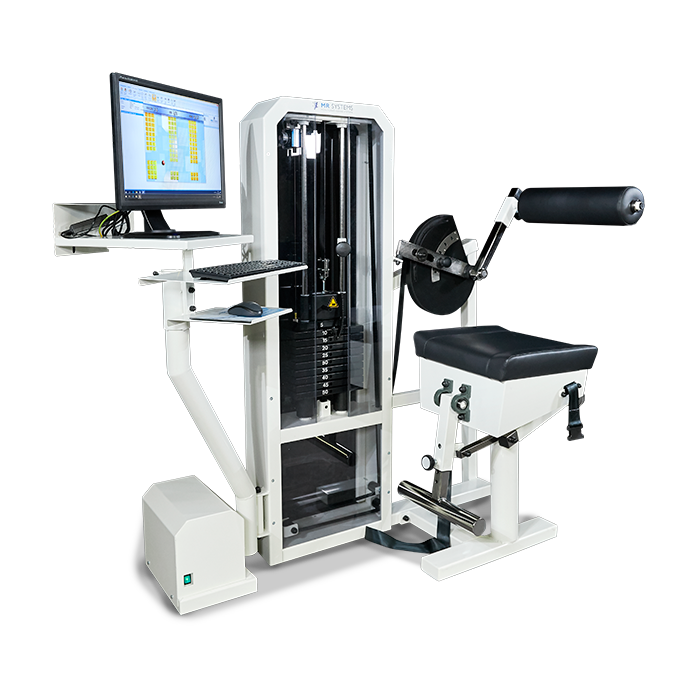
Cable Column
De Monitored Rehab Systems Cable Column is multifunctioneel en speciaal ontworpen voor optimale reconditionering, revalidatie en testen van de schouder en wervelkolom.
De unieke software bewaakt zowel motorische controle als reactieve neuromusculaire activiteiten die helpen om normale motorpatronen te herstellen.
Back Extension
De volledig instelbare hellingshoek van het bekken is een van de unieke kenmerken van de Monitored Rehab Systems Back Extension.
In combinatie met de mechanische ROM-begrenzer en de MRS Software is de Back Extension bij uitstek geschikt voor een breed scala aan therapeutische doelen.
Leg Extension
De Leg Extension met zijn revolutionaire ellipsvormige cam zorgt voor fysiologisch voldoende spierbelasting tijdens de volledige ROM en is daardoor uitermate geschikt voor patiënten met een beperkte belastbaarheid van de knie.
De Leg Extension helpt bij het opbouwen van kracht, balans, coördinatie en proprioceptie en zorgt tegelijkertijd voor optimale motivatie van de patiënt.
Cube
De MR Cube verhoogt onmiddellijk elk fysiotherapie- of atletisch conditieprogramma met realtime visuele feedback en begeleiding.
De MR Cube helpt bij het opbouwen van kracht, balans, coördinatie en proprioceptie en verbetert tegelijkertijd de therapietrouw van de patiënt. Interactieve videogames begeleiden de patiënt bij functionele oefeningen en geven duidelijke visuele feedback tijdens de therapie. Het resultaat is een ongeëvenaarde cognitieve betrokkenheid en verhoogde EMG-activiteit tijdens elke training.
De MR Cube is bijzonder geschikt voor het training met feedback op jouw bestaande trainingsapparatuur. De MR Cube kan binnen 2 seconden (met de bijgeleverde magneet) gekoppeld worden aan de jouw huidige trainingsapparatuur.
MR Functional Squat
De Monitored Rehab Systems Functional Squat is het eerste gesloten ketensysteem dat speciaal is ontworpen voor optimale reactieve neuromusculaire training en testen. Het unieke ontwerp van de draaiende slee repliceert de beweging van een staande squat tijdens het hele bewegingsbereik.
De Functional Squat is bij uitstek geschikt voor de functionele revalidatie van de onderste extremiteit van begin tot eindfase.

Medical Device Regulation
Met ingang van mei 2021 is de Verordening Medische Hulpmiddelen (MDR) van kracht. Deze Europese wetgeving stelt dat fabrikanten, importeurs en distributeurs van medische hulpmiddelen verplicht om hun processen in te richten conform deze Medical Device Regulation (MDR). Dit betekent onder andere dat de door hun ontwikkelende apparatuur geclassificeerd en gecertificeerd moet worden conform de eisen van deze wetgeving. Op dit moment voldoen de MR Functional Squat, MR Cube, MR Cable Column en de C-Mill aan de MDR eisen en zijn zodoende MDR gecertificeerd.
Meer informatie over onze apparatuur?
Neem contact met ons op en vraag naar de mogelijkheden.